There’s just no shortage of breakthroughs in the field of biomedicine these days. Whether it’s 3D bioprinting, bionics, nanotechnology or mind-controlled prosthetics, every passing week seems to bring more in the way of amazing developments. And given the rate of progress, its likely going to be just a few years before mortality itself will be considered a treatable condition.
There’s just no shortage of breakthroughs in the field of biomedicine these days. Whether it’s 3D bioprinting, bionics, nanotechnology or mind-controlled prosthetics, every passing week seems to bring more in the way of amazing developments. And given the rate of progress, its likely going to be just a few years before mortality itself will be considered a treatable condition.
Consider the most recent breakthrough in 3D printing technology, which comes to us from the J.B Speed School of Engineering at the University of Louisville where researchers used a printed model of a child’s hear to help a team of doctors prepare for open heart surgery. Thanks to these printer-assisted measures, the doctors were able to save the life of a 14-year old child.
 Philip Dydysnki, Chief of Radiology at Kosair Children’s Hospital, decided to approach the school when he and his medical team were looking at ways of treating Roland Lian Cung Bawi, a boy born with four heart defects. Using images taken from a CT scan, researchers from the school’s Rapid Prototyping Center were able to create and print a 3D model of Roland’s heart that was 1.5 times its actual size.
Philip Dydysnki, Chief of Radiology at Kosair Children’s Hospital, decided to approach the school when he and his medical team were looking at ways of treating Roland Lian Cung Bawi, a boy born with four heart defects. Using images taken from a CT scan, researchers from the school’s Rapid Prototyping Center were able to create and print a 3D model of Roland’s heart that was 1.5 times its actual size.
Built in three pieces using a flexible filament, the printing reportedly took around 20 hours and cost US$600. Cardiothoracic surgeon Erle Austin III then used the model to devise a surgical plan, ultimately resulting in the repairing of the heart’s defects in just one operation. As Austin said, “I found the model to be a game changer in planning to do surgery on a complex congenital heart defect.”
Roland has since been released from hospital and is said to be in good health. In the future, this type of rapid prototyping could become a mainstay for medical training and practice surgery, giving surgeons the options of testing out their strategies beforehand. And be sure to check out this video of the procedure from the University of Louisville:
And in another story, improvements made in the field of bionics are making a big difference for people suffering from diabetes. For people living with type 1 diabetes, the constant need to extract blood and monitor it can be quite the hassle. Hence why medical researchers are looking for new and non-invasive ways to monitor and adjust sugar levels.
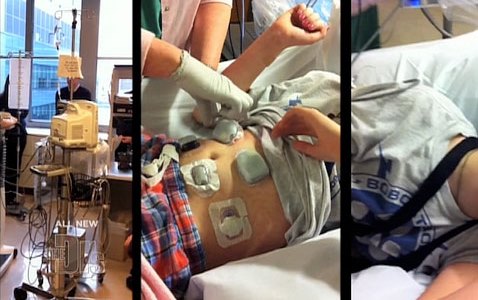
Solutions range from laser blood-monitors to glucose-sensitive nanodust, but the field of bionics also offer solutions. Consider the bionic pancreas that was recently trialled among 30 adults, and has also been approved by the US Food and Drug Administration (FDA) for three transitional outpatient studies over the next 18 months.
 The device comprises a sensor inserted under the skin that relays hormone level data to a monitoring device, which in turn sends the information wirelessly to an app on the user’s smartphone. Based on the data, which is provided every five minutes, the app calculates required dosages of insulin or glucagon and communicates the information to two hormone infusion pumps worn by the patient.
The device comprises a sensor inserted under the skin that relays hormone level data to a monitoring device, which in turn sends the information wirelessly to an app on the user’s smartphone. Based on the data, which is provided every five minutes, the app calculates required dosages of insulin or glucagon and communicates the information to two hormone infusion pumps worn by the patient.
The bionic pancreas has been developed by associate professor of biomedical engineering at Boston University Dr. Edward Damiano, and assistant professor at Harvard Medical School Dr. Steven Russell. To date, it has been trialled with diabetic pigs and in three hospital-based feasibility studies amongst adults and adolescents over 24-48 hour periods.
 The upcoming studies will allow the device to be tested by participants in real-world scenarios with decreasing amounts of supervision. The first will test the device’s performance for five continuous days involving twenty adults with type 1 diabetes. The results will then be compared to a corresponding five-day period during which time the participants will be at home under their own care and without the device.
The upcoming studies will allow the device to be tested by participants in real-world scenarios with decreasing amounts of supervision. The first will test the device’s performance for five continuous days involving twenty adults with type 1 diabetes. The results will then be compared to a corresponding five-day period during which time the participants will be at home under their own care and without the device.
A second study will be carried out using 16 boys and 16 girls with type 1 diabetes, testing the device’s performance for six days against a further six days of the participants’ usual care routine. The third and final study will be carried out amongst 50 to 60 further participants with type 1 diabetes who are also medical professionals.
 Should the transitional trials be successful, a more developed version of the bionic pancreas, based on results and feedback from the previous trials, will be put through trials in 2015. If all goes well, Prof. Damiano hopes that the bionic pancreas will gain FDA approval and be rolled out by 2017, when his son, who has type 1 diabetes, is expected to start higher education.
Should the transitional trials be successful, a more developed version of the bionic pancreas, based on results and feedback from the previous trials, will be put through trials in 2015. If all goes well, Prof. Damiano hopes that the bionic pancreas will gain FDA approval and be rolled out by 2017, when his son, who has type 1 diabetes, is expected to start higher education.
With this latest development, we are seeing how smart technology and non-invasive methods are merging to assist people living with chronic health issues. In addition to “smart tattoos” and embedded monitors, it is leading to an age where our health is increasingly in our own hands, and preventative medicine takes precedence over corrective.
Sources: gizmag.com, (2)